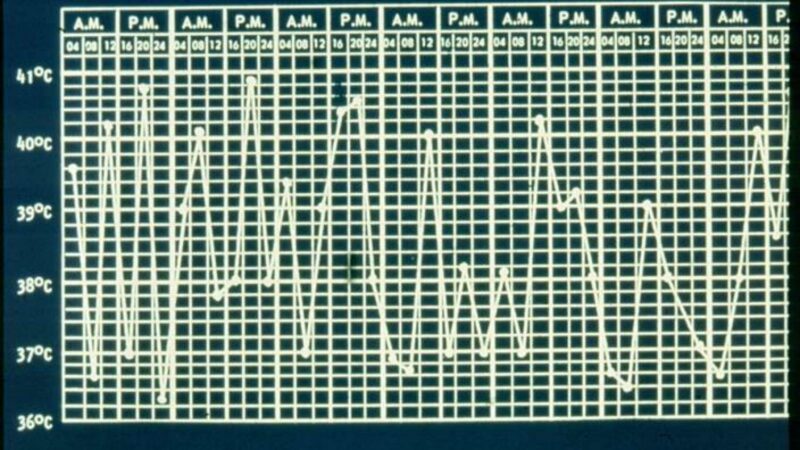
Althought EULAR currently recommends the pneumococcal vaccine be given to patients with inflammatory rheumatic diseases and the Centers for Disease Control and Prevention (CDC) recommends the vaccine in patients treated with immunosuppressive drugs, a recent brief report suggests it be avoided specifically in patients with cryopyrin-associated periodic syndrome or Behcet’s disease. (Citation source http://buff.ly/1nMjZC9)
This unusual phenomenon stems from an abstract submitted by Dr. Ulrich Walker, who heads up a canakinumab registry and has found at least 14 CAPS patients who had serious adverse events after receiving the vaccine. Patients ranged in age from 7 to 52 years old. Within a few hours of vaccination, all seven patients developed severe, local injection site reactions, fever and one case of presumed aseptic meningitis. Most received antibiotics, and two had to be hospitalized for systemic reactions. Symptoms resolved in 3-18 days.
The reactions occurred with pneumococcal vaccines from three different companies and in patients with different CAPS phenotypes (Muckle-Wells, NOMID, FCAS). Four vaccine reactions occurred in temporal association with concomitant co-injections of canakinumab; two reactions were separated by 15 days from the last canakinumab dose; and one reaction occurred in a patient who had never been exposed to canakinumab. Some patients had received other vaccines without complication.
These same investigators have also described a severe reaction to the PSV-23 valent vaccine in a Behcet’s patient (http://buff.ly/1PWPHJK).
It is unclear if such hyperreactivity will be seen with IL-1 inhibitors or other autoinflammatory syndrome patients or those with systemic JIA or adults with Still’s disease. Caution and further study is warranted.
Related Content
-
October 10, 2016
The diagnosis of periodic fevers is unified by undiagnosed but recurrent fever. Unfortunately…
-
August 1, 2016
Tumor necrosis factor (TNF) receptor-associated periodic syndrome (TRAPS) is an autosomal-dominant autoinflammatory disease…
-
May 14, 2022
Dr. Jack Cush reviews the appproach to evaluating fever in adults suspected of…
-
October 15, 2018
Some clinical benefits were seen among patients with adult-onset Still's disease treated with…
-
January 1, 1970
A retrospective records review of patients with periodic fever syndromes (PFS) receiving IL-1…
-
March 13, 2019
First-line treatment of systemic juvenile idiopathic arthritis (JIA) with anakinra (Kineret) was highly…