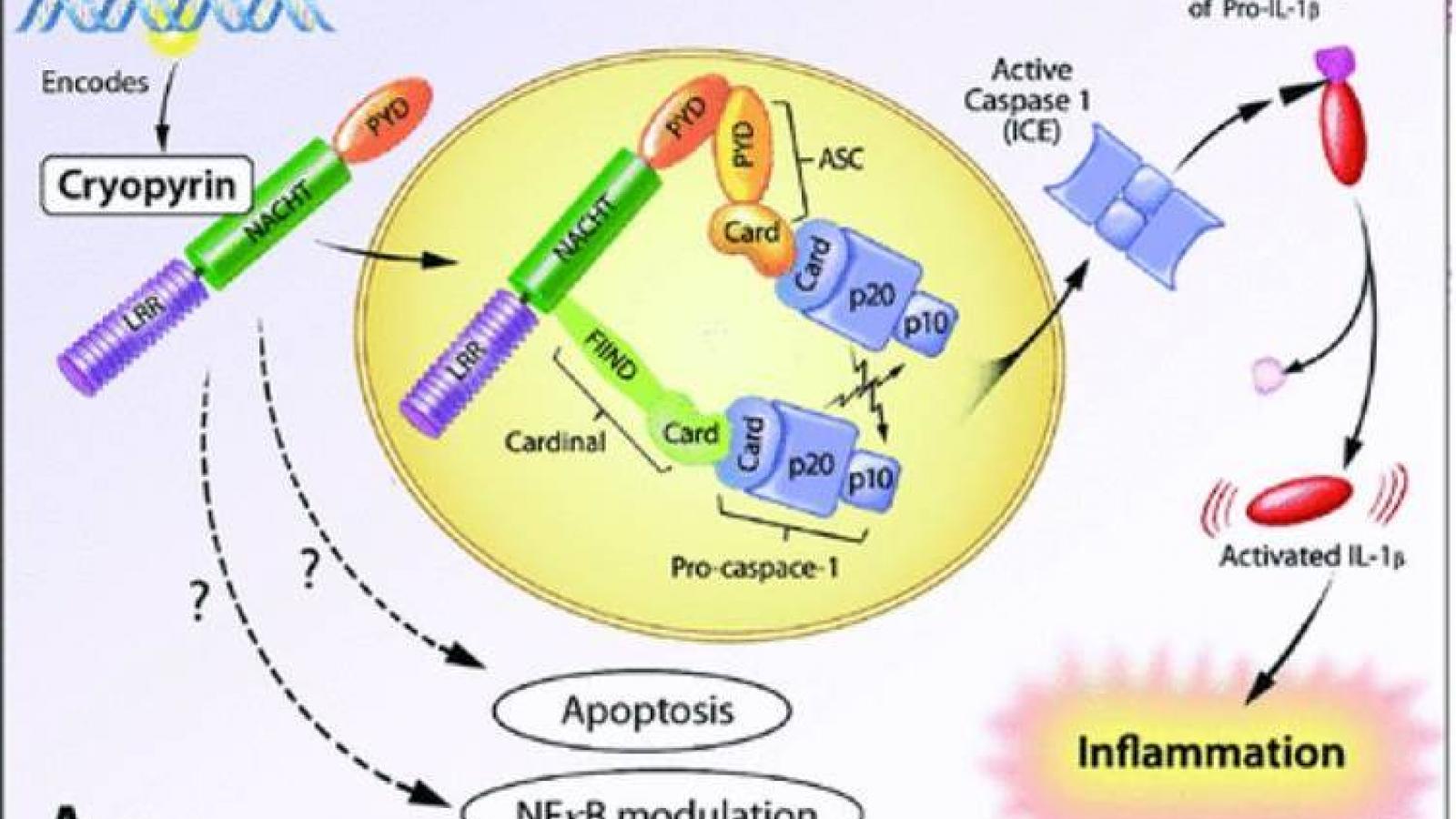
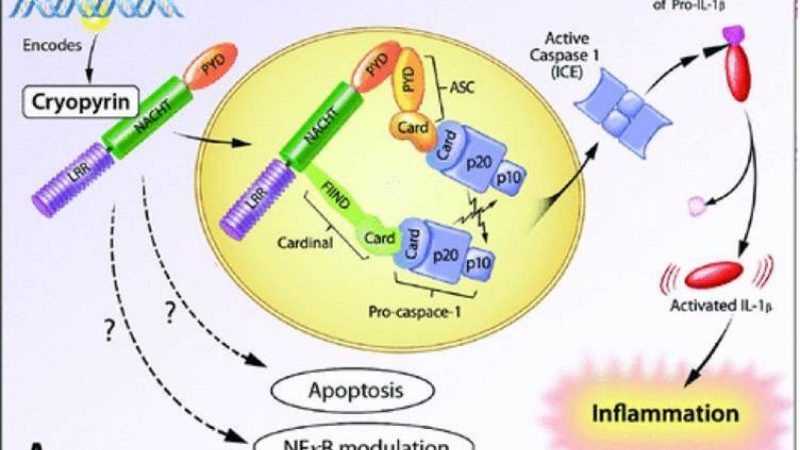
Autoinflammatory syndromes manifest as periodic fevers in children, and are the result of monogenic, “gain in function” gene alterations affecting the inflammasome. The innate immune response is driven by inflammasomes, which are cytosolic multiprotein complexes in macrophages that respond to stimuli (i.e., pathogens, stress, urate), activating a caspase-1–mediated cascade that results in an excess of interleukin-1 and the generation of an inflammatory response.
The gene mutations underlying inflammasome activity have been well described, but there appears to be variable penetrance among patients with the same gene mutation, suggesting additional mechanisms may influence disease expression.
Researchers from the Bellvitge Biomedical Research Institute examined the role of DNA demethylation in activating inflammasome genes during macrophage differentiation and monocyte activation in healthy control subjects and patients with cryopyrin-associated periodic syndromes (CAPS) and familial Mediterranean fever (FMF). CAPS patients have mutations in the encoding gene cryopyrin, but not all CAPS patients have the same clinical features or respond equally to treatment. (Citation source: http://buff.ly/29XyyMq)
In normal controls, inflammasome genes are demethylated during monocyte differentiation and activation, and this results in increased gene expression. When patients with autoinflammatory syndromes were examined, the monocytes from untreated CAPS patients had more efficient DNA demethylation than healthy controls. Moreover their DNA methylation reverts back to control levels with the use of IL-1 inhibitors.
These findings suggest that epigenetic changes (DNA methylation) involving the inflammasome may be used therapeutically or as novel biomarkers in studying patients with CAPS or FMF.
Related Content
-
November 10, 2020
With broader awareness about the disease state, early diagnosis and treatment in autoinflammatory…
-
January 1, 1970
Saturday's session on autoinflammatory syndromes featured Dr. Dan Kastner addressing - The Adult…
-
January 1, 1970
This podcast features a discussion of autoinflammatory and Still's disease abstracts from virtual…
-
June 11, 2020
-
May 27, 2020
This week's Tuesday Nite Rheumatology featured Dr Randy Cron from the University of Alabama…
-
May 14, 2022
Vexed about VEXAS: Dr. Janet Pope https://youtu.be/hfm54-5IZLQ Diagnosing VEXAS Syndrome https://youtu.be/KOec2hXUBeU …