Only inhibitors of IL-1 (canakinumab) and IL-6 (tocilizumab) are FDA approved for use in children with systemic onset juvenile idiopathic arthritis (sJIA). Now, long-term data from the extension studies from two phase III studies shows that canakinumab yields long-term improvements with reduced glucocorticoid dosing with no new safety findings with long-term use.
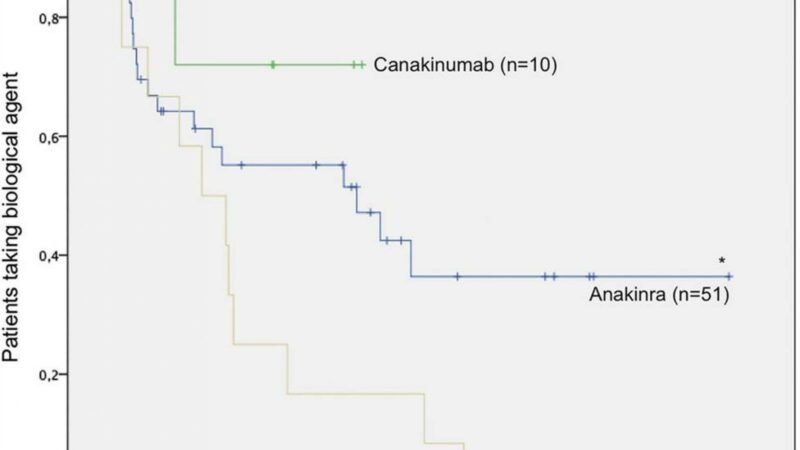
Nearly 80% of the 177 patients enrolled in the core study entered the open label extension, and of these, 75 patients (42%) completed the 5-year study. The remaining 102 (58%) discontinued mainly for inefficacy.
Discontinuation rates noted were higher in late responders (81%) compared with early responders (29%) to canakinumab.
At 2 years, aJIA-ACR 50/70/90 response rates were 62%, 61% and 54%, respectively.
Clinical remission was achieved by 20% of patients at month 6; 32% at 2 years. A JADAS low disease activity score was achieved by 49% of patients at 2 years.
A total of 128/177 (72.3%) patients were on glucocorticoids at entry and 15.6% discontinued and 22% tapered steroids to 0.150 mg/kg/day.
Overall the safety profile of canakinumab remained unchanged. Serious adverse events included flares of sJIA (25 events or 5.24/100 patient-years) and 13 patients with macrophage activation syndrome (17 events or 3.56/100 patient-years) and there were no new deaths.
One limitation of this study may be the number of dropouts – with 58% of the patients discontinuing canakinumab over the 5 years. This may be explained by the very long duration of the LTE and the fact that some patients with sJIA will spontaneously remit.
Related Content
-
February 20, 2019
The drug retention rate of interleukin-1 inhibitors (IL-1) used to treat systemic juvenile…
-
May 5, 2015
A retrospective review of 77 SoJIA patients revealed that 50-70% achieved inactive disease or remission…
-
August 20, 2020
You can reduce - but not stop - canakinumab therapy in patients who…
-
January 1, 1970
A retrospective records review of patients with periodic fever syndromes (PFS) receiving IL-1…
-
November 9, 2021
-
October 12, 2020