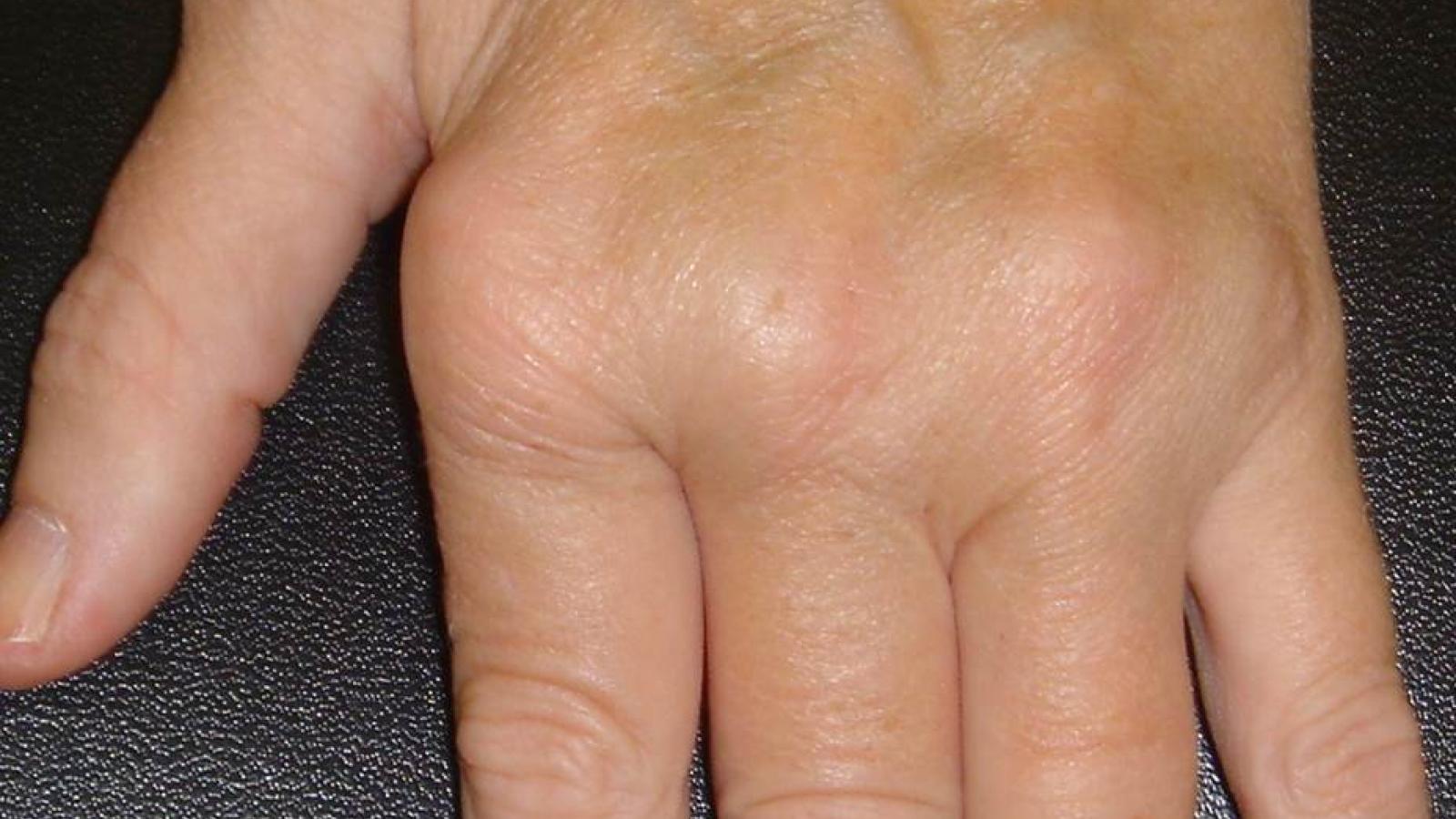
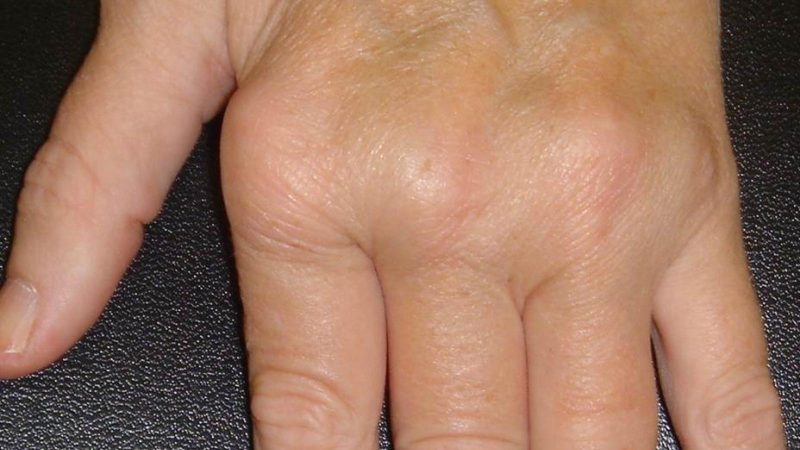
Yesterday, the FDA approved canakinumab (Ilaris) as treatment for adult-onset Still’s disease (AOSD), the first ever FDA approved drug for AOSD.
Canakinumab, an interleukin-1 inhibitor, was FDA approved in 2013 for use children (aged 2 years and older) with systemic juvenile idiopathic arthritis (sJIA) and has since been approved for use in other periodic fever disorders (e.g., FMF, CAPS, TRAPS, Hyper IgD syndrome).
Ilaris was granted priority review designation by the FDA and has approved canakinumab for AOSD based on clinical trial data and the drugs prior record in sJIA and other periodic fevers. Dosing and use can soon be found on the drugs website. IL-1 inhibition has been shown to be effective in managing both the articular and systemic features of Still’s disease.
Common side effects with Ilaris are infections (colds and upper respiratory tract infections), abdominal pain and injection-site reactions. The prescribing information for Ilaris includes a warning for potential increased risk of serious infections due to IL-1 blockade.
By this approval the FDA has tacitly affirmed that AOSD and sJIA are essentially the same disorder, differing only in the onset age.
Related Content
-
February 17, 2020
-
November 8, 2021
-
February 12, 2021
-
September 8, 2017
Adult-onset Still's disease (AOSD) is the adult continum of systemic-onset juvenile idiopathic arthritis…
-
February 26, 2018
Gabay and colleagues have reported the results of a novel new recombinant human…
-
February 13, 2020