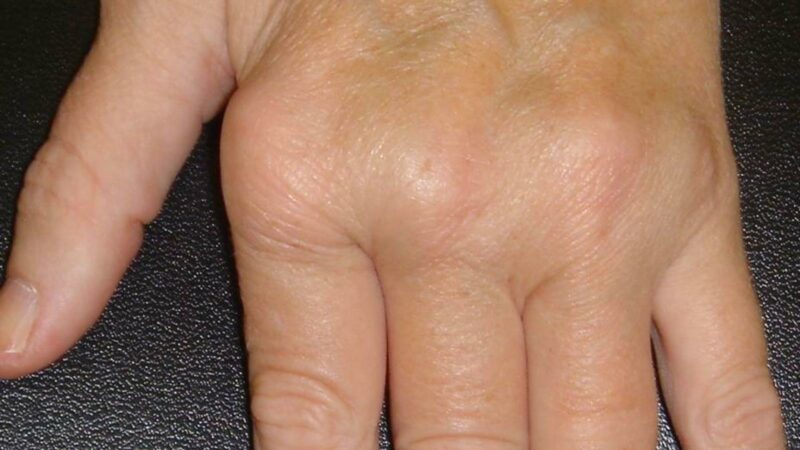
Adult-onset Still’s disease (AOSD) is the adult continum of systemic-onset juvenile idiopathic arthritis (SoJIA). Treatment of this febrile polyarticular systemic disorder can be complex, but has been relieved by several specific biologic therapies.
Currently there are no drugs (including biologics) specifically approved for use in AOSD, yet there are several that approved for use in SoJIA – indicated for use in active Systemic Juvenile Idiopathic Arthritis (SJIA) in patients aged 2 years and older:
Approved Biologics for SoJIA
- Ilaris (canakinumab)
- Actemra (tocilizumab)
Biologics Not Approved (but occassionally used)
- Arcalyst (rilonacept) is an interleukin-1 blocker
- Kineret (anakinra): is an IL-1 receptor antagonist
Biologics Under Study (according to clinicaltrials.gov)
- Ilaris (canakinumab) – under study for AOSD
- Kineret (anakinra): under study for AOSD and SoJIA
A recent systematic review of the literature identified those biologic drugs that have been studied in AOSD. The meta-analysis included 19 observational studies and showed overall rate of clinical response of 0.85 (95% CI: 0.77-0.91, p < 0.0001) and an overall rate of complete remission of 0.66 (95% CI: 0.54-0.77, p = 0.01). These studies were hampered by a high rate of heterogeneity (Q=59.82 with df=19.0,p<0.0001,I2=68.23%).
Most of these results are driven by the success of IL-1 or IL-6 inhibition in controlling the systemic features of Still’s disease – including fever, rash, hepatosplenomegaly, lymphadenopathy, arthritis, serositis and aggressively inflammatory indices.
TNF inhibitors are NOT effective enough to warrant use in those with active inflammatory systemic disease, but may be considered in those whose disease has settled into a dominant rheumatoid-like chronic, polyarthritis.
Related Content
-
February 17, 2020
-
November 8, 2021
-
February 12, 2021
-
June 17, 2020
Yesterday, the FDA approved canakinumab (Ilaris) as treatment for adult-onset Still's disease (AOSD),…
-
February 26, 2018
Gabay and colleagues have reported the results of a novel new recombinant human…
-
February 13, 2020