Since 2000, the German Biologics register (BiKeR) has prospectively enrolled children with juvenile idiopathic arthritis to assess outcomes with biologic therapies. A new report evaluates the efficacy and safety of etanercept (ETA), tocilizumab (TOC) and the interleukin-1 inhibitors (anakinra and canakinumab) (IL-1i) in those with systemic onset juvenile idiopathic arthritis JIA (sJIA).
Currently, only TOC and canakinumab are FDA approved for use in sJIA patients over age 2 years in the USA.
Overall they evaluated 245 sJIA patients (50.3% male) exposed to biologics.
- 143 patients treated with ETA
- 71 with TOC
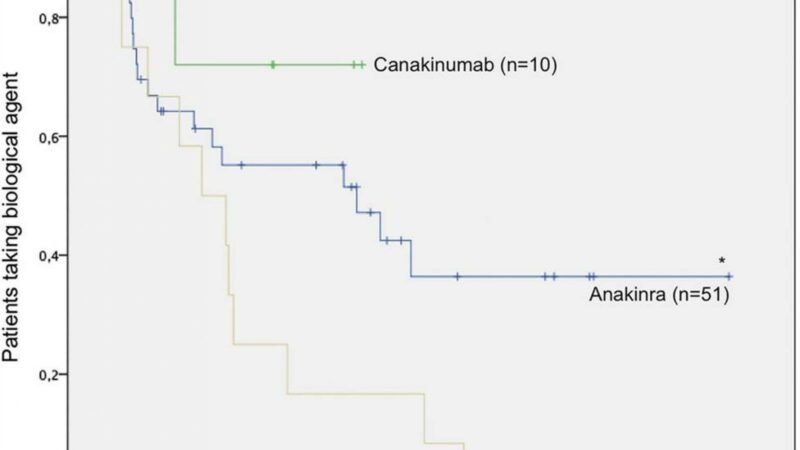
- 60 with IL-1i (anakinra 38, canakinumab 22).
Concomitant treatment with systemic steroids was less frequent with TOC and IL-1i than with ETA for .
The ETA cohort had fewer systemic disease manifestations (1.4% vs 42.3% vs 63% ETA/TOC/IL1i) but more active joints.
IL-1i and TOC cohorts achieved better JIA-ACR 30/50/70/90 responses after 24 months than ETA. Also, prior therapy with another biologic did not significantly alter subsequent biologic responses.
24 Month Results
| ETA | TOC | IL-1i | |
| JADAS remission | 20% | 37% | 52% |
| Minimal Dz activity | 35% | 61% | 68% |
| ACR inactive disease | 24% | 33% | 56% |
Serious adverse events were observed more frequently with TOC (RR 2.5; p < 0.5) and IL1i (2.9; p < 0.01), but such patients manifested more systemic findings at the outset.Serious infections were rare but were numerically higher with IL-1i. Lastly, 11 cases of macrophage activation syndrome (MAS) occurred with all 3 biologics; 3 with ETA,, 5 with TOC and 3 in the IL-1i cohorts.
TOC or IL-1i therapy is effective in sJIA, with JADAS remission reached by up to half of patients while up to two thirds reached JADAS minimal disease activity.
ETA is clearly less effective in systemic JIA and its use has markedly decreased in Germany. ETA is effective in managing the inflammatory arthritis of sJIA, but not the systemic aspects of sJIA.
Related Content
-
April 5, 2018
The German Autoinflammatory Disease (AID) registry has studied the effects of the IL-6…
-
May 5, 2015
A retrospective review of 77 SoJIA patients revealed that 50-70% achieved inactive disease or remission…
-
November 26, 2019
A disease you’ve never heard of is becoming increasingly common and carries a…
-
March 13, 2019
First-line treatment of systemic juvenile idiopathic arthritis (JIA) with anakinra (Kineret) was highly…
-
August 8, 2019
A single-center cohort analysis shows that lung disease (LD) is increasingly seen in…
-
January 1, 1970
Systemic onset juvenile idiopathic arthritis (sJIA) is a dramatic onset inflammatory condition marked…